Your best source on science and technology news from Washington state
Provided by AGPOpus Genetics Completes Enrollment in Cohort 1 of Phase 1/2 OPGx-BEST1 Gene Therapy Study
Target Enrollment Achieved with the Inclusion of Participants with Both Dominant and Recessive Forms of BEST Disease
Baseline Demographics Presented at ARVO Annual Meeting with Summary Audio and Slide Presentation Available on Opus Website
Three-Month Topline Results from Cohort 1 Expected in September 2026
RESEARCH TRIANGLE PARK, N.C., May 07, 2026 (GLOBE NEWSWIRE) -- Opus Genetics, Inc. (Nasdaq: IRD) (“Opus Genetics” or the “Company”), a clinical-stage biopharmaceutical company developing gene therapies to restore vision and prevent blindness in patients with inherited retinal diseases (IRDs), today announced the completion of enrollment in Cohort 1 of its ongoing Phase 1/2 study of OPGx-BEST1 gene therapy.
The adaptive, open-label study is evaluating the safety and efficacy of single-eye subretinal administration of OPGx-BEST1 up to two dose levels in adult participants with Best Vitelliform Macular Dystrophy (BVMD) or Autosomal-Recessive Bestrophinopathy (ARB). Five participants have been enrolled in the study, three with BVMD and two with ARB. The first four participants have been dosed and the fifth participant is scheduled for the dosing procedure this month.
“As planned, we enrolled a mix of participants with the dominant and recessive forms of BEST disease to evaluate OPGx-BEST1 in both conditions,” said George Magrath, M.D., Chief Executive Officer, Opus Genetics. “We collaborated closely with our study sites and investigators to identify participants who met our carefully defined entry criteria. In the dominant participants, we completed the added step of using an in vitro platform to confirm that each participant’s disease mutation is amenable to gene augmentation. The promising tolerability data and early efficacy signals we saw in the sentinel participant show the promise for individuals living with this potentially blinding disease. We are grateful to the participants and clinical sites who are participating in the trial, and we look forward to presenting the three-month data on the full cohort later this year.”
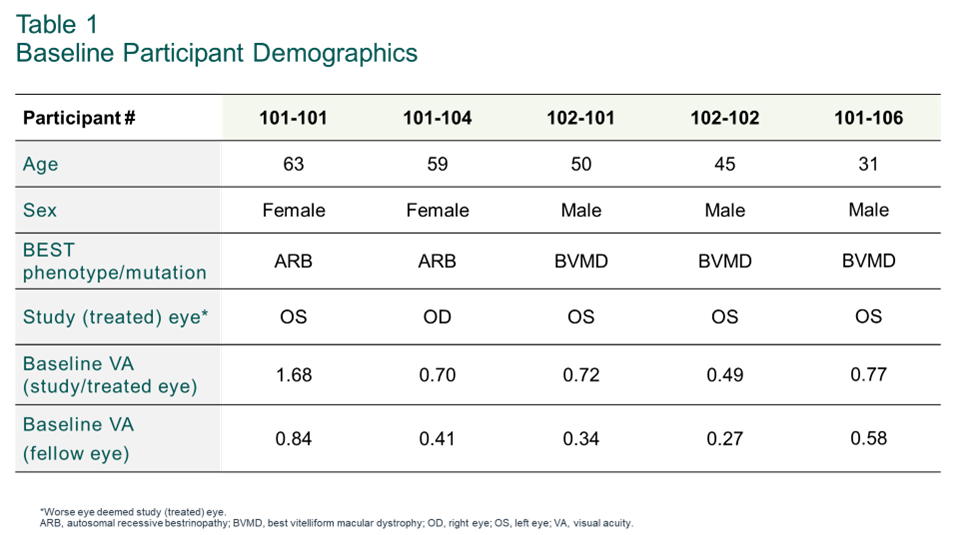
In a session today at the Association for Research in Vision and Ophthalmology (ARVO) Annual Meeting, the study’s principal investigator, Mark Pennesi, M.D., Ph.D., Chief Medical Officer and Director, Inherited Retinal Degeneration Clinic, Retina Foundation of the Southwest, presented baseline demographics of Cohort 1 (summarized in Table 1) and 3-month results from the first (sentinel) participant treated in the study, highlighting positive tolerability and biological activity following subretinal administration of OPGx-BEST1.
The ARVO poster presentation and a video recording of Dr. Pennesi’s summary of Cohort 1 Baseline Demographics and Key Endpoints for IRDs are available on the Opus Genetics website in the Events section.
Opus expects to announce 3-month topline data from Cohort 1 in September 2026, followed by the presentation of data at an ophthalmology medical conference later this year. Data is expected to be provided on multiple outcome measures including structural optical coherence tomography (OCT) measures, microperimetry, best‑corrected visual acuity, low luminance visual acuity and contrast sensitivity.
About OPGx-BEST1
OPGx-BEST1 leverages Opus Genetics’ proprietary AAV-based gene therapy platform, designed to deliver a functional copy of the BEST1 gene directly to the retinal pigment epithelium (RPE) cells where the defective gene resides. The program builds on extensive preclinical work demonstrating restoration of BEST1 protein expression and improved retinal function in relevant disease models.
By restoring BEST1 function, the therapy aims to address the underlying genetic cause of retinal degeneration and support preservation of photoreceptor health and visual function. BEST1-associated IRDs affect an estimated 22,000 patients worldwide and currently have no approved treatments.
About Opus Genetics
Opus Genetics is a clinical-stage biopharmaceutical company developing gene therapies to restore vision and prevent blindness in patients with inherited retinal diseases (IRDs). The Company is developing durable, one-time treatments designed to address the underlying genetic causes of severe retinal disorders. The Company’s pipeline includes seven AAV-based programs, led by OPGx-LCA5 for LCA5-related mutations and OPGx-BEST1 for BEST1-related retinal degeneration, with additional candidates targeting RDH12, MERTK, RHO, CNGB1 and NMNAT1. Opus Genetics is based in Research Triangle Park, NC. For more information, visit www.opusgtx.com.
Forward Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Such statements include, but are not limited to, statements related to the clinical development, clinical results, preclinical data, and future plans for OPGx-BEST1 and expectations regarding us, our business prospects, and our results of operations and are subject to certain risks and uncertainties posed by many factors and events that could cause our actual business, prospects and results of operations to differ materially from those anticipated by such forward-looking statements. Factors that could cause or contribute to such differences include, but are not limited to, those described under the heading “Risk Factors” included in our Annual Report on Form 10-K for the fiscal year ended December 31, 2025, our subsequent Quarterly Reports on Form 10-Q, and in our other filings with the U.S. Securities and Exchange Commission. Readers are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this press release. These forward-looking statements are based upon our current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties. In some cases, you can identify forward-looking statements by the following words: “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “aim,” “may,” “ongoing,” “plan,” “potential,” “predict,” “project,” “should,” “strive,” “will,” “would” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. We undertake no obligation to revise any forward-looking statements in order to reflect events or circumstances that might subsequently arise.
Contacts:
Investors
Jenny Kobin
Remy Bernarda
IR Advisory Solutions
ir@opusgtx.com
Media
Kimberly Ha
KKH Advisors
917-291-5744
kimberly.ha@kkhadvisors.com
Source: Opus Genetics, Inc.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/c8c9006e-0278-4c21-a78c-6da68677c067

Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.


